Ready-to-administer Calcium Gluconate in non-PVC, non-DEHP containers is designed to promote safety and convenience
Fresenius Kabi announced today it has introduced two new presentations of Calcium Gluconate in Sodium Chloride Injection in the United States in the company’s proprietary, ready-to-administer freeflex® containers.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20220427005262/en/
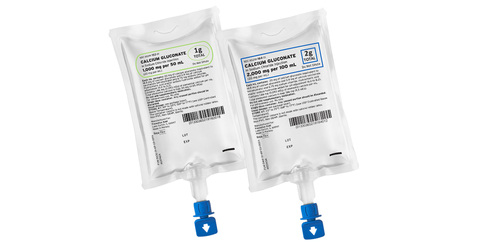
Ready-to-administer Calcium Gluconate in non-PVC, non-DEHP containers from Fresenius Kabi. (Photo: Business Wire)
Fresenius Kabi now has the broadest portfolio of Calcium Gluconate Injection products available from any manufacturer in the United States:
- Calcium Gluconate in Sodium Chloride Injection freeflex in 1,000 mg per 50 mL
- Calcium Gluconate in Sodium Chloride Injection freeflex in 2,000 mg per 100 mL
- Calcium Gluconate Injection Single Dose Vial in 1,000 mg per 10 mL
- Calcium Gluconate Injection Single Dose Vial in 5,000 mg per 50 mL
- Calcium Gluconate Injection Pharmacy Bulk Package in 10,000 mg per 100 mL
“Our new premixed Calcium Gluconate freeflex offerings will help meet the growing demand for ready-to-administer medicines in the U.S.,” said John Ducker, president and CEO of Fresenius Kabi USA. “By offering Calcium Gluconate Injection in two delivery systems, Fresenius Kabi can provide health care professionals with more flexibility and choice to best serve patient needs.”
freeflex is an innovative, non-PVC and non-DEHP, multilayer polyolefin film bag designed for infusion solutions that has been marketed globally since 2005 and in the U.S. since 2008. freeflex promotes the safety and convenience of infusion therapy in a ready-to-administer presentation.
Indications and Usage
- Calcium Gluconate in Sodium Chloride Injection is a form of calcium indicated for pediatric and adult patients for the treatment of acute symptomatic hypocalcemia.
- Limitations of Use: The safety of Calcium Gluconate Injection for long term use has not been established.
Important Safety Information
Calcium Gluconate in Sodium Chloride Injection is contraindicated in hypercalcemia and in neonates (28 days of age or younger) receiving ceftriaxone.
Arrhythmias with Concomitant Cardiac Glycoside Use: If concomitant therapy is necessary, Calcium Gluconate in Sodium Chloride Injection should be given slowly in small amounts and close ECG monitoring is recommended.
End-Organ Damage due to Intravascular Ceftriaxone-Calcium Precipitates: Concurrent use of intravenous ceftriaxone may cause life-threatening precipitates. Do not administer ceftriaxone simultaneously with Calcium Gluconate in Sodium Chloride via a Y-site in any age group. Cases of fatal outcomes in neonates have occurred.
Tissue Necrosis and Calcinosis: Calcinosis cutis can occur with or without extravasation of Calcium Gluconate in Sodium Chloride Injection. Tissue necrosis, ulceration, and secondary infection are the most serious complications. If extravasation occurs or clinical manifestations of calcinosis cutis are noted, immediately discontinue intravenous administration at that site and treat as needed.
Hypotension, Bradycardia, and Cardiac Arrhythmias with Rapid Administration: To avoid adverse reactions that may follow rapid intravenous administration, Calcium Gluconate in Sodium Chloride Injection should be infused slowly, with careful ECG monitoring for cardiac arrhythmias.
Aluminum Toxicity: This product contains aluminum, up to 100 mcg per liter, that may be toxic.
The most common adverse events with Calcium Gluconate Injection are local soft tissue inflammation and necrosis, calcinosis cutis and calcification that are related to extravasation. Other adverse events include vasodilation, decreased blood pressure, bradycardia, cardiac arrhythmia, syncope, and cardiac arrest.
To report SUSPECTED ADVERSE REACTIONS, contact Fresenius Kabi USA, LLC at 1-800-551-7176, option 5, or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions:
Cardiac Glycoside: Synergistic arrhythmias may occur if calcium and cardiac glycosides are administered together.
Calcium Channel Blockers: Administration of calcium may reduce the response.
Drugs that may cause hypercalcemia: Vitamin D, vitamin A, thiazide diuretics, estrogen, calcipotriene and teriparatide administration may cause hypercalcemia. Monitor plasma calcium concentrations in patients taking these drugs concurrently.
Geriatric use: Dosing in elderly patients should be cautious usually starting at the low end of the dosage range.
Renal impairment: Initiate with the lower limit of the dosage range and monitor serum calcium levels every 4 hours.
This Important Safety Information does not include all the information needed to use Calcium Gluconate in Sodium Chloride Injection safely and effectively. Please see full prescribing information for Calcium Gluconate in Sodium Chloride Injection at www.fresenius-kabi.com/us.
About Fresenius Kabi
Fresenius Kabi (www.fresenius-kabi.com/us) is a global health care company that specializes in medicines and technologies for infusion, transfusion, and clinical nutrition. The company’s products and services are used to help care for critically and chronically ill patients. The company’s U.S. headquarters is in Lake Zurich, Illinois. The company’s global headquarters is in Bad Homburg, Germany. To learn about U.S. career opportunities at Fresenius Kabi, visit us at http://www.fresenius-kabi.com/us/join-us.
View source version on businesswire.com: https://www.businesswire.com/news/home/20220427005262/en/
Contacts
Joanie Clougherty (614) 717-5741
joan.clougherty@fresenius-kabi.com





