- Developing NRX-101, an FDA-Designated Investigational Breakthrough Therapy for Suicidal Treatment-Resistant Bipolar Depression and Chronic Pain.
- Designed to Help Address the Needs of Over 13 Million Americans who Seriously Consider Suicide Each Year (CDC).
- D. Boral Analyst Report on NRXP $34 Price Target.
- Type C Meeting with the FDA Demonstrates a Path to New Drug Application with Real World Data and Broader Proposed Indication for NRX-100 (ketamine).
- Strategic Partnership with Emobot to Integrate AI-Powered “Depression Thermometer” Across Interventional Psychiatry Network
- 2025 Results Included Reduction in Operating Expenses with $7.8 Million Cash on Hand. Adequate Resources to Support Operations Through 2026.
- FDA Bioequivalence Determination by Office of Generic Drugs for NRx’s Preservative-Free Ketamine Application.
- Anticipated FDA Approval of at Least One NRXP New Drug – Preservative Free Ketamine Under an ANDA—During Q3 2026.
- Opening of Palm Beach, FL Clinic for One Day Treatment for Depression and PTSD, Now Accepting Patients.
- Published Results Have Shown 87% Clinical Response to Non-Invasive Transcranial Magnetic Stimulation with Neuroplastic Medications.
- Clinical Leadership by Physicians Trained at Harvard, Johns Hopkins, Georgetown, and Other Leading Universities.
- Prof. Joshua Brown MD, PhD, Appointed as Chief Medical Innovation Officer.
- In-Person Meeting attended with FDA Division of Psychiatry Products and Leadership of the FDA Center for Drug Evaluation and Research (CDER).
- On Path to Application for New Drug Approval of NRX-100 Under Fast Track Designation Based on Existing Clinical Trial Data and Real World Evidence.
- NRXP Will Seek Broader Indication to Serve Patients with Treatment-Resistant Depression Who May Have Suicidality Rather Than the Subset with Suicidality.
- 70,000 Patient Data on Real World Use of Ketamine for Treatment of Suicidal Depression to be Submitted to the FDA in Support of NRX-100 Approval
- Analyses Suggested Clinical Response is Consistent with Prior Randomized Trial Data and Compares Favorably to Currently-Approved Products.
- NRXP and neurocare Group AG Announce Joint Offering of Neuroplastic Therapy Targeting Depression, PTSD and Other Mental Health Afflictions.
- Elimination of All Balance Sheet Debt Following Equity Conversion.
- New Pipeline Indication for Augmentation of Transcranial Magnetic Stimulation.
- Applied for Use of KETAFREE™ as a Proprietary Product Name as First Preservative-Free Ketamine Formulation.
- Current Worldwide Generic Ketamine Market Estimated at $750 Million Per Year.
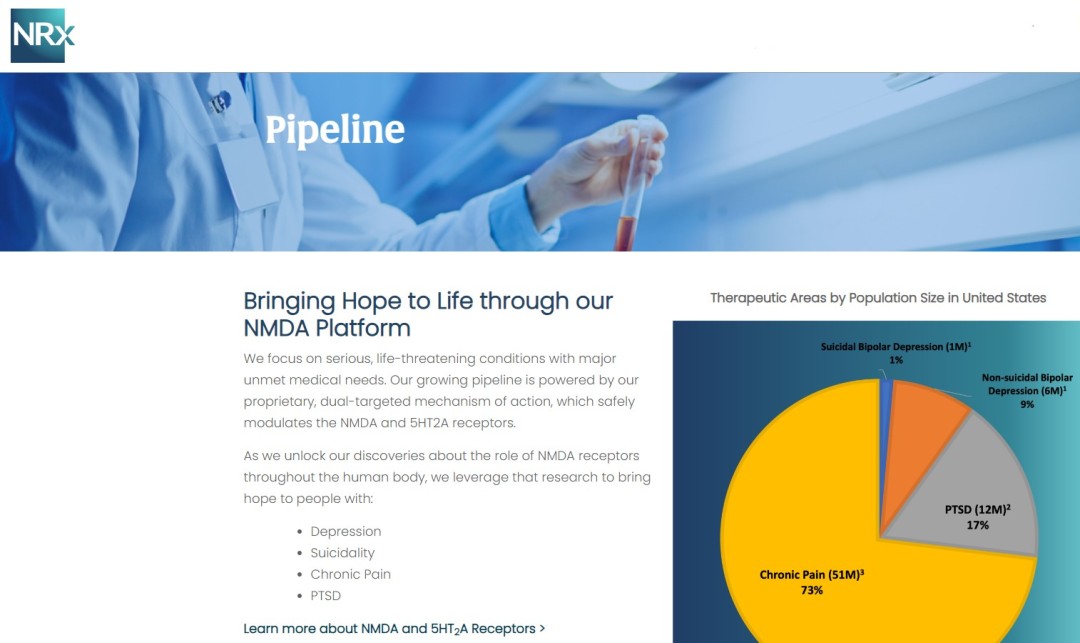
NRx Pharmaceuticals, Inc. (Nasdaq: NRXP) is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. NRXP is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain
NRXP has partnered with Alvogen Pharmaceuticals around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRXP is working on a New Drug Application for NRX-100 (IV ketamine) in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRXP was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
D. Boral has issued an Analyst Report on NRXP with a Buy and $34 Price Target. The full report may be accessed at this direct link: https://www.nrxpharma.com/wp-content/uploads/2025/11/HOPE-Therapeutics-NRXP-Executes-Florida-Roll-out-of-Ampa-O.pdf
Strategic Partnership with Emobot to Integrate AI-Powered “Depression Thermometer” Across Interventional Psychiatry Network
On March 30th NRXP announced a strategic partnership with Emobt Health to deploy its revolutionary AI-driven emotional monitoring platform across the rapidly expanding NRXP network of interventional psychiatry clinics. This roll-out represents the first large-scale clinical integration of Emobot’s "Depression Thermometer," a passive monitoring tool designed to detect early treatment response and prevent relapse in patients with Treatment-Resistant Depression (TRD).
Emobot’s technology replaces cumbersome psychometric questionnaires with a background application on the user’s smartphone to passively and automatically measure levels of depression and potentially suicidality and PTSD. In recent years, there has been widespread recognition that neuroplastic therapies, including potentially IV Ketamine, SPRAVATO® (esketamine), and Transcranial Magnetic Stimulation (TMS) have the potential to replace an older and less effective generation of oral antidepressants.
Full-Year 2025 Results and Highlights Key Regulatory and Commercial Progress
On March 24th NRXP announced the filing of its Form 10-K for the year ended December 31, 2025. Highlights included the following key points:
A year-over-year reduction in operating expenses with $7.8 Million cash on hand at year end. With ongoing revenue from operations and current ATM trends, NRXP anticipates adequate resources to support operations through 2026, with anticipation of continued revenue growth. As of December 31, 2025, all previously issued convertible debt had been converted into common stock.
Anticipated FDA approval of at least one NRXP new drug – Preservative Free Ketamine under an ANDA—during Q3 2026. A favorable preliminary bioequivalence determination was received from the FDA.

A Type C in-person meeting with the FDA Division of Psychiatry Products together with leadership of the FDA Center for Drug Evaluation and Research (CDER) to identify a path to approval of NRXP NRX-100 (preservative-free ketamine) under a New Drug Application with an indication for treatment of Severe Depression in Patients who may have suicidality. In meeting minutes, FDA expressed willingness to review existing randomized clinical trial data and Real World Evidence as a potential basis for approval without a requirement for conduct of additional clinical trials.
Appointment of Prof. Joshua Brown, MD, PhD, of Harvard/Mclean as NRXP Chief Medical Innovation Officer. Dr. Brown is Principal Investigator on NIH- and DARPA-funded projects that highlight the future of neuroplastic care, including use of D-cycloserine and TMS for treating depression, PTSD, and suicidality.
Filing of an Investigational New Drug application with the FDA to study the use of the NRXP Breakthrough-designated candidate, NRX-101, for the enhancement of Transcranial Magnetic Stimulation (TMS) supported by non-dilutive governmental funding sources.
Development of a patentable, sustained-release presentation of D-cycloserine to provide an extended release profile suitable for enhancement of TMS efficacy. Prior NRXP clinical trials have shown a doubling of clinical response in patients with depression and an 8-fold increase in remission from depression versus standard TMS therapy.
Implementation of a business plan for NRXP subsidiary HOPE Therapeutics, Inc. with the aim of developing interventional psychiatry centers focused on neuroplastic therapy – including ketamine, D-cycloserine, Transcranial Magnetic Stimulation, and Hyperbaric Oxygen Therapy – for patients with severe and suicidal depression and PTSD, with future applications for Traumatic Brain Injury, Autism, and Alzheimer’s.
First NRXP revenue from acquired and partnered interventional psychiatry clinics at five sites of care that provide neuroplastic therapy to treat severe and suicidal depression and PTSD, with funding from the Department of Veterans Affairs, the Department of War, private insurers, and self-pay. Company expects to substantially increase this revenue-generating footprint in 2026.
Announcement of a nationwide NRXP partnership with neurocare AG, a leading manufacturer of TMS technology to develop their growing combined clinic base, currently at 20 sites of care in the US together with an installed base of more than 400 TMS machines into a nationwide accountable care network providing psychiatrist-led neuroplastic therapy.
FDA Bioequivalence Determination by Office of Generic Drugs for NRx’s Preservative-Free Ketamine Application
On March 17th NRXP announced that it has received a letter from the Bioequivalence Program of the FDA Office of Generic Drugs stating that “FDA has not identified any bioequivalence deficiencies at this time.” The determination is deemed preliminary until final supervisory review of the NRXP Abbreviated New Drug Application with anticipated approval in Summer 2026.
Opening of Palm Beach, FL Clinic for One Day Treatment for Depression and PTSD, Now Accepting Patients
On March 9th NRXP announced the opening of its Palm Beach, FL clinic location dedicated to the treatment of Depression and PTSD with an interventional psychiatry approach that includes ketamine1 and other neuroplastic drugs, transcranial magnetic stimulation (TMS), and hyperbaric oxygen, combined with physician-led psychotherapy. This NRXP clinic location is the most recent addition to HOPE’s growing network of clinics.
Peer-reviewed scientific publications have shown an 87% clinical response rate with a short-term treatment protocol in patients with treatment-resistant depression. With the addition of hyperbaric oxygen therapy, NRXP pilot programs have shown a 90% rate of return to full function in patients with depression and post-traumatic stress disorder. TMS has also been shown in some studies to demonstrate improved memory and cognition in some patients with Alzheimer’s disease and Traumatic Brain Injury. NRXP previously announced a nationwide partnership with neurocare AG and expects to bring the latest developments in the non-invasive treatment of these conditions to this flagship location.
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Email: mduffy@nrxpharma.com
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Media Contact
Company Name: NRx Pharmaceuticals, Inc.
Contact Person: Matthew Duffy, Chief Business Officer
Email: Send Email
Phone: 484 254 6134
Address:1201 Orange Street Suite 600
City: Miami
State: Florida
Country: United States
Website: https://www.nrxpharma.com/







